Describe the Process Used to Electroplate a Material
The electrodes are immersed in an electrolyte a solution. All of these items can be purchased at a local hardware or home improvement store.
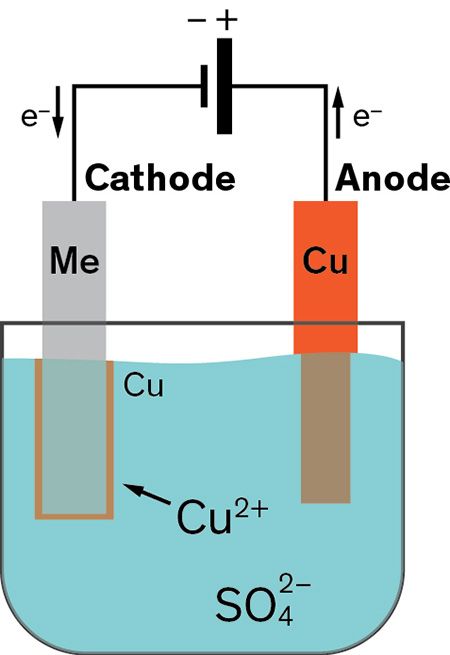
Copper Electroplating Fundamentals
Describe the electroplating process.
. The electroplating process is an application of an electrolytic cell that requires two different metals an electrolyte solution two electrodes and a battery or other power source that generates an electric current. Metal plating provides many benefits to products made from metal and other materials. A cell consists of two electrodes conductors usually made of metal which are held apart from one another.
How it works The. This is mostly achieved through electroplating which requires an electric current or through electroless plating which is in autocatalytic chemical process. This is useful for coating a cheaper metal with a more expensive one such as copper or silver.
The electrolyte is a solution of a salt of the metal to be coated. In addition it is also common for objects to arrive at the shop with old metal coatings. Engineers use controlled electrolysis to transfer the desired metal coating from an anode a part containing the metal that will be used as the plating to a cathode the part to be plated.
Electro nickel plating also known as nickel electroplating or nickel electro-deposition is becoming an increasingly popular process for a variety of different manufacturing applications. Gather the necessary materials. Electroplating is mainly done for avoiding corrosion of metals and for decorative purposes.
Its primarily used to increase wear resistance protect against corrosion increase thickness or change the aesthetic appeal of an object. An electrical current passing through the solution causes objects at the cathode to be coated by the metal in the solution. Using electricity you can coat the metal of one electrode with the metal of the other in an electroplating process also known as electrochemistry.
Disassembly Dissembling all of the connected parts ensures there is an even coat of electroplating solution on all of the surface area. This process can be used to give objects increased wear resistance corrosion protection or aesthetic appeal as well as increased thickness. In the word Electroplating electro refers to electric current and plating refers to an act of outer covering of some materials with a thin layer of a different metal.
Describe the electroplating process. Electrolysis is used to electroplate objects coat them with a thin layer or metal. For example metals like iron are coated with deposits of nickel or chromium by electroplating to protect it from corrosion.
In electroplating a more-expensive metal is plated deposited in a thin layer onto the surface of a cheaper metal by electrolysis. Electroplating Makes Use of an Electrical Current. The electroplating process also uses a non-metallic coating to protect the surface from corrosion and make it corrosion resistance.
Classify vapour deposition processes into physical vapour deposition PVD and chemical vapour deposition CVD. Electroplating uses a form of electrolysis in which the electrodes play a bigger role than just conducting the current. To begin determine which anode and cathode and electrolyte to use by determining which chemical reaction or reactions you want.
Stripping Removes particles which can blister or flake a plate-layer. The electroplating process create a layer of other material on the surface of the substrate to be protected this smoothness the surfaces and prevent any sort of tarnishing corrosion or wear and tear. Explain the process of anodizing and state typical applications.
This process results in a thin layer of metal being deposited onto the surface of a workpiece called the substrate. Electroplating is the process of applying a metallic coating to an article by passing an electric current through an electrolyte in contact with the article thereby forming a. A List typical plating materials and give reasons for their use.
The electroplating is an art of depositing a superior or more noble metal on a base metal by means of electrolysis. Electroforming is a metal forming process in which parts are fabricated through electrodeposition on a model known in the industry as a mandrel. Thus electroplating is defined as the process of plating a layer of any metal on other material by passing an electric current.
The plating process is a manufacturing process in which a thin layer of metal coats a substrate. When the electricity flows through the circuit they make the electrolyte splits up and some of the metal atoms it contains are deposited in a thin layer on top of one of the electrodesit becomes electroplated. Electroplating is the application of electrolytic cells in which a thin layer of metal is deposited onto an electrically conductive surface.
Electroplating is a process by which metal ions migrate via a solution from a positive electrode to a negative one. Electroplating is the process of using electrodeposition to coat an object in a layer of metals. All kinds of metals can be plated in this way including gold silver tin zinc copper cadmium chromium nickel platinum and lead.
And the anode positive electrode is usually either a. Electrolytic cells are also used in a process called electroplating. Electroplating is primarily used to change the physical properties of an object.
Electro nickel plating is a process that uses an electrical current to coat a conductive material typically made of metal with a thin layer of nickel. To electroplate with this method you will need a piece of copper the metal to be plated vinegar hydrogen peroxide alligator clips a 6-volt lantern battery a glassplastic container and gloves. Before plastic auto bumpers became popular chromium metal was electroplated onto steel bumpers.
Conductive metallic mandrels are treated to create a mechanical parting layer or are chemically passivated to limit electroform adhesion to the mandrel and thereby allow its subsequent separation. The electroplating process uses an electric current to deposit a thin layer of material on top of an object. Electroplating is a general name for processes that produce a metal coating on a solid substrate through the reduction of cations of that metal by means of a direct electric currentThe part to be coated acts as the cathode negative electrode of an electrolytic cell.
The Electroplating Process Broken Down Into 8 Steps
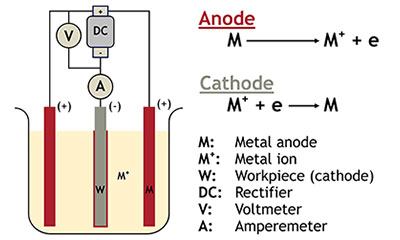
Electroplating Process How Does It Work Its Process And Applications
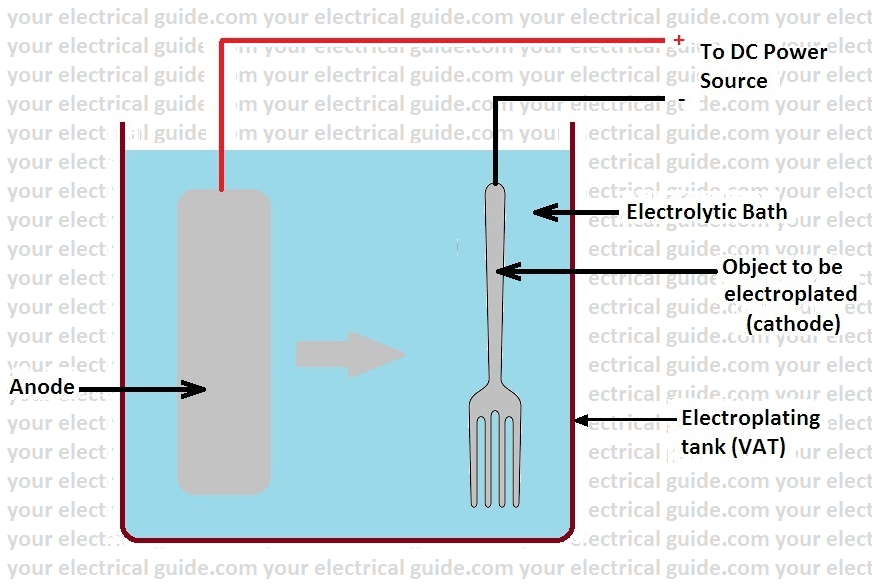
Electroplating Process Steps Your Electrical Guide

What Is Electroplating Definition Working Principle Its Uses
Comments
Post a Comment